Elective surgery without COVID-19 testing will lead to excess morbidity and mortality
ABSTRACT: Countless surgical procedures have been canceled worldwide due to the COVID-19 pandemic. As surgical volumes increase globally to address unmet surgical need, consideration must be given to how to navigate surgical risk during this pandemic. Using current COVID-19 prevalence rates, the risk of operating on COVID-19–infected patients in the absence of routine mandatory testing was modeled. Assuming 37 000 planned surgeries are conducted per week in Canada (excluding Quebec), and the prevalence of COVID-19 infection cases is 0.20%, the number of avoidable deaths that could occur is estimated to be 11.7 but could exceed 17.0. Given the risk of increased morbidity and mortality after elective surgery in asymptomatic COVID-19-infected patients, preoperative testing should be considered mandatory.
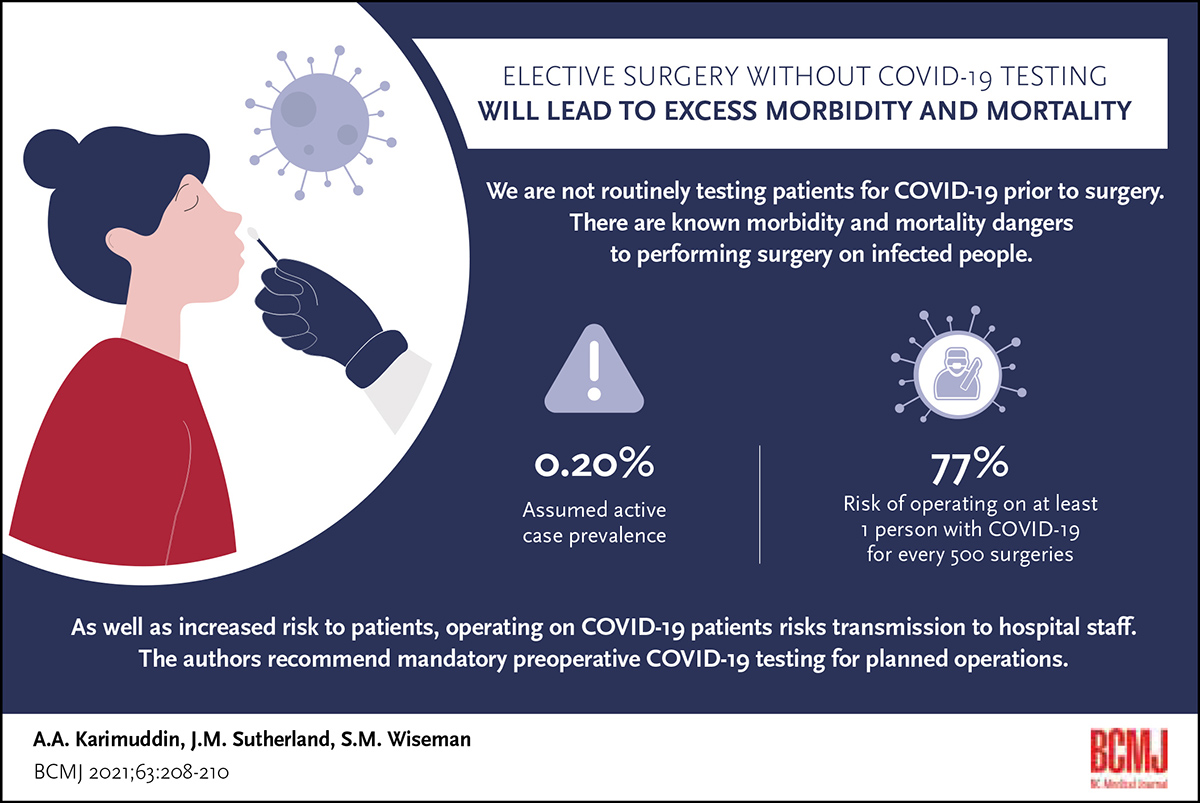
Patients should be tested for COVID-19 prior to planned operations to prevent avoidable surgical complications and mortality.
Based on global experiences during the early days of the COVID-19 pandemic, most elective operations were canceled to create capacity for an anticipated surge in pandemic-related hospitalizations. Cancelation of elective surgeries reduced the risk of hospitals serving as transmission sites and avoided a significantly increased risk of postoperative complications and death.[1,2]
As the pandemic begins to ebb in many countries, there is a focus on returning to prepandemic surgical volumes, and even increasing volumes to catch up with delayed or canceled procedures.[3] Whether in public health care systems, where surgical access was previously constrained and now is further exacerbated by the pandemic, or in mixed public–private health care models like those in the United States, where providers view surgical services as a business, there will be increasing demands on surgical capacity and a need for increasing surgical volumes.[4,5]
Preoperative testing
Faced with pressure to increase surgical volumes, hospitals should understand their options and the risks of inaction regarding COVID-19 infection. The first decision is whether to preoperatively test all asymptomatic elective surgical patients for infection. The feasibility and utility of testing varies from centre to centre and is influenced by many factors, including the test itself (availability, practicality, performance metrics), type of proposed operation (risk of aerosolization), preferences of the surgical team (surgeons, anesthesiologists, and nurses), centre-specific characteristics (policies, facility size, surgical volumes, availability of personal protective equipment), and surgical population (age, comorbidities, COVID-19 prevalence rate).
Recent research has raised concerns about increased risks of pulmonary complications and mortality in COVID-19–infected patients, whether they are asymptomatic or presymptomatic at the time of elective surgery.[6,7] Although many infected patients are completely asymptomatic or have a few minor symptoms, they have significantly increased perioperative morbidity and mortality. Furthermore, they pose an infection risk to all hospital personnel and other patients they come into contact with during their hospitalization. Recently, the Misericordia Community Hospital in Alberta had to be closed and all elective surgeries were canceled for a second time due to a hospital-wide COVID-19 outbreak.
Direction from public health, specifically in Canadian centres, has not included mandatory COVID-19 testing of asymptomatic patients who are considered low risk on screening prior to elective surgery.[8] While prescreening can identify some people who are at increased risk of harboring COVID-19 infection and warrant preoperative testing, it has not been shown to be effective or to protect patients or their health care providers when compared with mandatory testing. Routine preoperative testing of elective surgical patients has been recommended in guidelines issued by many surgical organizations.[9,10] At the height of the pandemic in New York City, 99 orthopaedic surgical patients were screened and also underwent nasopharyngeal swab testing for COVID-19. Seven of 12 patients (58.3%) who were found to be infected with COVID-19 were completely asymptomatic; however, even the asymptomatic patients suffered from a higher incidence of postsurgical pulmonary complications than would otherwise be expected.[11] Based on similar observations, researchers from Italy concluded that surgery should be postponed in COVID-19–infected patients whenever possible.[12] Thus, the possibility of patients undergoing elective surgery when unknowingly infected with COVID-19 represents a critically important preoperative concern.
Modeling risk
We modeled the likelihood of operating on a COVID-19–infected patient based on assumptions regarding COVID-19 prevalence in the population. Current prevalence data from Canada and other countries, which ranged from a low of 0.01% to a high of 0.30% prevalence, were used to form the model’s assumptions. These rates reflect current findings from member countries of the Organisation for Economic Co-operation and Development, whereas according to the Worldometer, as of 12 January 2021, the prevalence of active cases was 0.20% in Canada and 2.74% in the United States. At a local active case prevalence of 0.20%, for every 500 surgeries conducted in a hospital, the risk of operating on at least one person with COVID-19 is 77%.
According to the Canadian Institute for Health Information, between 1 February and 30 April 2019, approximately 475 000 operations, or 37 000 per week, were performed in Canada, excluding Quebec. While it is not known what proportion of these planned operations were considered emergent and had to be performed regardless of patient COVID-19 infection status, based on our assumption of a 0.20% active case prevalence in Canada and extrapolating from 2019 Canadian surgical volumes, more than 70 patients with COVID-19 are likely to be operated on weekly.
COVID-19 has been shown to significantly increase morbidity and mortality among surgical patients.[6] Fifty-three percent of COVID-19–infected patients are assumed to have pulmonary complications, and their death rate is assumed to be 25%; among COVID-19–infected patients who do not experience pulmonary complications, the death rate is assumed to be 9%. These outcomes are compared with the assumption that 15% of operative cases have pulmonary complications, and pulmonary complication-related deaths account for 5%, whereas the death rate among patients without pulmonary complications is 1%. The difference between the two models represents the “excess” deaths attributable to increased morbidity and mortality of COVID-19–infected patients who have undergone surgery.
The Figure illustrates the excess, or avoidable, deaths in a non-testing environment, assuming 37 000 planned surgeries per week in Canada (excluding Quebec). Assuming the prevalence of COVID-19 infection cases is 0.20%, the number of excess deaths is estimated to be 11.7. It is important to note that the number of excess deaths is an underestimation because surgical case volumes for Quebec were not included, and COVID-19 preoperative testing methodologies are currently evolving and may not diagnose all infected cases. Further, if the true Canadian prevalence rate is higher (e.g., 0.40%) due to variable testing practices or a lack of testing among asymptomatic Canadians, it is expected that the number of excess deaths will exceed 23.5. The impact of vaccination, and the presence of viral variants, on perioperative COVID-19–related mortality and morbidity, and whether it should influence preoperative testing protocols, is not established and warrants further study.
Summary
Government policymakers, hospitals, surgeons, other medical personnel, and patients must reckon with the knowledge that even at the lowest COVID-19 prevalence rates, without preoperative testing, infected people will undergo elective surgery and have an increased risk of avoidable morbidity and mortality; they will also present an avoidable risk of transmission to hospital staff.
Currently, no province has a mandate to routinely test elective surgical patients for COVID-19 preoperatively. Based on our analyses, we recommend that provinces and hospitals mandate preoperative testing for planned operations to prevent avoidable surgical complications and patient mortality.
Competing interests
None declared.
This article has been peer reviewed.
References
1. Aminian A, Safari S, Razeghian-Jahromi A, et al. COVID-19 outbreak and surgical practice: Unexpected fatality in perioperative period. Ann Surg 2020;272:e27-e29.
2. Li Y-K, Peng S, Li L-Q, et al. Clinical and transmission characteristics of Covid-19 – A retrospective study of 25 cases from a single thoracic surgery department. Curr Med Sci 2020;40:295-300.
3. British Columbia Ministry of Health. A commitment to surgical renewal in BC. Coronavirus COVID-19. 7 May 2020. Accessed 25 February 2021. www2.gov.bc.ca/assets/gov/health/conducting-health-research/surgical-renewal-plan.pdf.
4. Kruse FM, Jeurissen PPT. For-profit hospitals out of business? Financial sustainability during the COVID-19 epidemic emergency response. Int J Heal Policy Manag 2020;9:423-428.
5. Nepogodiev D, Omar OM, Glasbey JC, et al. Elective surgery cancellations due to the COVID-19 pandemic: Global predictive modelling to inform surgical recovery plans. Br J Surg 2020;107:1440-1449.
6. COVIDSurg Collaborative. Mortality and pulmonary complications in patients undergoing surgery with perioperative SARS-CoV-2 infection: An international cohort study. Lancet 2020;396:27-38.
7. Nahshon C, Bitterman A, Haddad R, et al. Hazardous postoperative outcomes of unexpected COVID-19 infected patients: A call for global consideration of sampling all asymptomatic patients before surgical treatment. World J Surg 2020;44:2477-2481.
8. British Columbia Centre for Disease Control, British Columbia Ministry of Health. Infection prevention and control (IPC) protocol for surgical procedures during COVID-19: Adult. Coronavirus COVID-19. 21 May 2020. Accessed 25 February 2021. www.bccdc.ca/Health-Professionals-Site/Documents/COVID19_IPCProtocolSurgicalProceduresAdult.pdf.
9. Dowdy S, Fader AN. Surgical considerations for gynecologic oncologists during the COVID-19 pandemic. Society of Gynecologic Oncology. Accessed 25 February 2021. www.sgo.org/wp-content/uploads/2020/03/Surgical_Considerations_Communique.v14.pdf.
10. American College of Surgeons. Local resumption of elective surgery guidance. 17 April 2020. Accessed 25 February 2021. www.facs.org/covid-19/clinical-guidance/resuming-elective-surgery.
11. Gruskay JA, Dvorzhinskiy A, Konnaris MA, et al. Universal testing for COVID-19 in essential orthopaedic surgery reveals a high percentage of asymptomatic infections. J Bone Joint Surg Am 2020;102:1379-1388.
12. Doglietto F, Vezzoli M, Gheza F, et al. Factors associated with surgical mortality and complications among patients with and without Coronavirus disease 2019 (COVID-19) in Italy. JAMA Surg 2020;155:1-14.
Dr Karimuddin is a surgeon in the Department of Surgery, St. Paul’s Hospital, and a clinical associate professor at the University of British Columbia. Dr Sutherland is a professor at the Centre for Health Services and Policy Research, School of Population and Public Health, University of British Columbia. Dr Wiseman is a surgeon in the Department of Surgery, St. Paul’s Hospital, and a professor at the University of British Columbia.

