Common pitfalls in the diagnosis and management of bacterial infections
A review of the literature to determine how clinicians may fall victim to cognitive traps when working through the diagnostic and therapeutic process, with guidance on how to avoid these pitfalls when managing adult patients with suspected infections.
Infections are one of the most common conditions encountered in both inpatient and outpatient settings, with infections of the respiratory tract, urinary tract, and integumentary system frequently diagnosed.[1-3] Proving the presence of an infection is not usually possible in routine clinical practice, because a biopsy of the affected tissue or organ is needed to demonstrate histological invasion and the destruction of host cells by pathogens. Therefore, medical practitioners rely on history, physical exam findings, and laboratory tests to determine whether an infection might exist. This data-gathering process is akin to the work of a detective putting together pieces of the puzzle to solve a diagnostic mystery. Sometimes the puzzle pieces don’t fit perfectly with each other, irrelevant or distracting information clouds the clinical picture, or not all of the data are available for a clinician to be 100% confident about the diagnosis. The threshold at which diagnostic certainty impacts a clinical decision varies from person to person. Some might be comfortable proceeding with treatment when they are only 50% confident about the diagnosis, while others might need more certainty before doing so. Unlike the diagnosis of malignancies, where tissue biopsy (the gold standard) is required to confirm the disease before prescribing chemotherapy, the diagnosis of common bacterial infections is typically made without confirmation when antibiotic treatment is initiated.
If we cannot prove with a tissue biopsy that an infection is present, how often do we get the diagnosis wrong? And, consequently, how often are we mistreating patients with antibiotics? I will consider these questions using three common infections as examples: respiratory tract infections, urinary tract infections, and skin and soft tissue infections. These examples and the studies referenced apply to adult patients only.
Respiratory tract infections
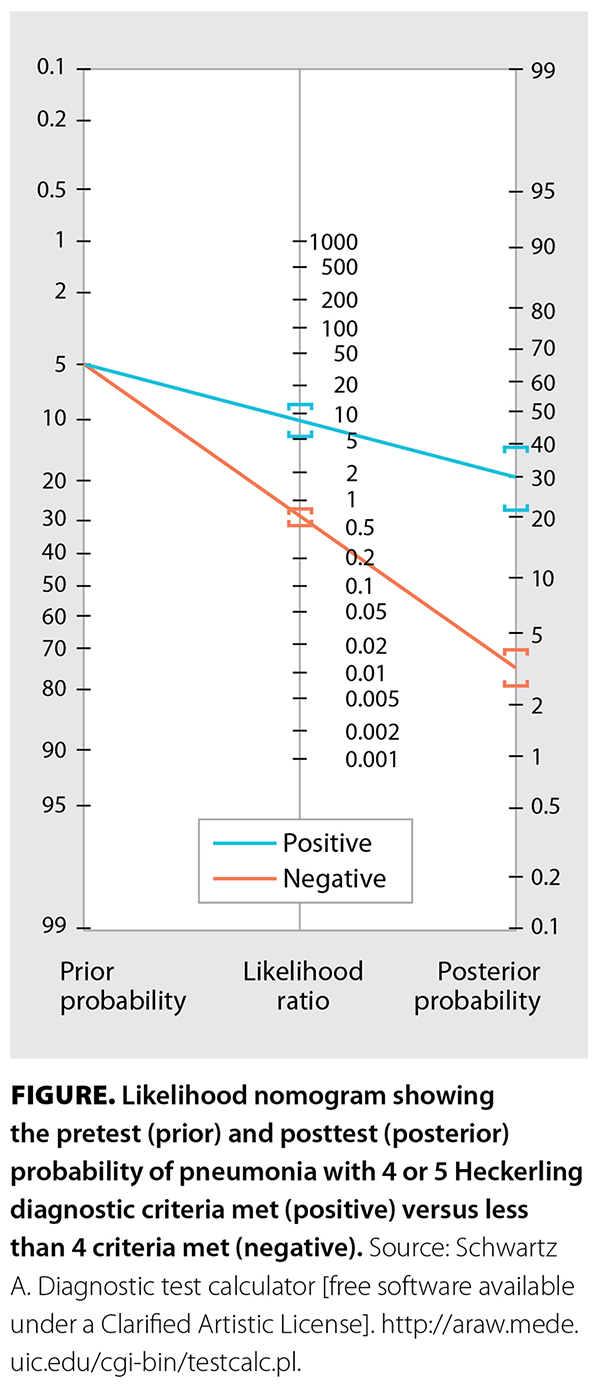 Consider this scenario: “A previously healthy 35-year-old woman who smokes tobacco presents with 5 days of fatigue, productive cough, worsening shortness of breath, temperatures to 38.9 °C, and decreased breath sounds in the lower right field. She has a heart rate of 105 beats/minute, but vital signs are otherwise normal.”[4] What is the probability that she has pneumonia? In this survey study (N = 553), nearly all respondents overestimated the probability at a median of 80%,[4] while the evidence-based answer is roughly 30% using the Heckerling diagnostic score [Figure], assuming the pretest probability of pneumonia is 5% for a patient with a cough.[5,6] What is the probability if the chest X-ray is either positive or negative? The median answers were 95% and 50%, respectively, while the evidence-based estimates are roughly 55% and 15%, respectively.[4]
Consider this scenario: “A previously healthy 35-year-old woman who smokes tobacco presents with 5 days of fatigue, productive cough, worsening shortness of breath, temperatures to 38.9 °C, and decreased breath sounds in the lower right field. She has a heart rate of 105 beats/minute, but vital signs are otherwise normal.”[4] What is the probability that she has pneumonia? In this survey study (N = 553), nearly all respondents overestimated the probability at a median of 80%,[4] while the evidence-based answer is roughly 30% using the Heckerling diagnostic score [Figure], assuming the pretest probability of pneumonia is 5% for a patient with a cough.[5,6] What is the probability if the chest X-ray is either positive or negative? The median answers were 95% and 50%, respectively, while the evidence-based estimates are roughly 55% and 15%, respectively.[4]
In all scenarios, respondents grossly overestimated the probability of pneumonia, uncovering a fundamental weakness with diagnostic reasoning and test interpretation. Due to the presence of cognitive biases that can impair probabilistic and statistical reasoning, including base rate neglect, anchoring bias, and confirmation bias, humans have a tendency to overestimate, which results in diagnostic errors.[4] Overdiagnosis subsequently leads to overtreatment.
The most common tool we depend on to diagnose pneumonia is chest X-ray, but how reliable is it? Unfortunately, its sensitivity and specificity are mediocre, at 69% and 78%, respectively.[7] Given the inter-observer variability in physical exam findings and the imperfect accuracy of chest X-ray, it is not surprising that pneumonia is often misdiagnosed, with 34% of outpatients being labeled with it when they don’t have it.[8]
Antibiotic treatment for pneumonia is also prone to errors. In one study of outpatients with community-acquired pneumonia (N = 341), only 31% received a guideline-concordant regimen.[8] Treatments were deemed inappropriate due to the wrong choice of drug (77%) and/or the wrong duration—almost always too long (39%). Lack of awareness about the microbial etiology of pneumonia might be tied to selection of the wrong drug. According to data from the United States, the most frequently detected pathogens in community-acquired pneumonia are viruses (22%), followed by bacteria (11%).[9] Because differentiating between viral and bacterial pneumonia can be challenging, I wonder what proportion of viral infections are being treated inappropriately with antibiotics.
It is also important to recognize the limitations of microbiological testing. A nasopharyngeal swab is performed to detect viruses, while a sputum culture is sometimes obtained to identify bacteria. Caution must be exercised when interpreting a positive result. Viral testing employs a nucleic acid amplification technique, which cannot distinguish between viable and dead organisms.[10] Moreover, identifying a virus from the nasopharynx does not necessarily prove causation of pneumonia or exclude a bacterial infection. Sputum cultures also have limited utility, because isolating the culprit bacteria from a nonsterile site is difficult. Further complicating the matter, for positive sputum cultures, determining whether the organism is pathogenic or simply a colonizer is problematic.[10] For these reasons, guidelines recommend against obtaining routine sputum cultures in the management of community-acquired pneumonia, except in patients hospitalized with severe infection or if there is concern for drug-resistant pathogens.[11] However, the isolation of certain organisms, including some atypical bacteria (e.g., Nocardia), fungi (e.g., Cryptococcus), and mycobacteria (e.g., Mycobacterium tuberculosis), is almost always considered pathogenic and warrants further investigation and treatment.
Urinary tract infections
Let’s start with a clinical case. “A 65-year-old man is seen for osteoarthritis. He has noted foul-smelling urine but no pain or difficulty with urination. A urine dipstick shows trace blood.”[4] What is the probability of a urinary tract infection (UTI)? In this survey, the median estimated pretest probability by participants was 20%.[4] If a urine culture is either positive or negative, respondents predicted a median chance of UTI of 80% and 5%, respectively.[4] Foul-smelling urine does not count as a UTI symptom, and, by definition, asymptomatic bacteriuria is not a UTI, so the evidence-based probability is essentially 0% regardless of the urine culture result. Not only did clinicians overcall the probability, but the impact of a positive urine culture on their estimate was also alarmingly high.
It is intriguing that medical practitioners have such strong faith in a positive urine culture as robust evidence for a UTI. It appears that the concept of asymptomatic bacteriuria is poorly understood, as evidenced by studies that found that 71% of respondents would prescribe unnecessary antibiotic for this condition[12] and 83% of hospitalized patients with asymptomatic bacteriuria received inappropriate antibiotic.[13]
Dysuria, urinary frequency, hematuria, back or lower abdominal pain, and absence of vaginal discharge or irritation are the only evidence-based UTI symptoms.[14] An exception is the isolation of Staphylococcus aureus from urine culture, which might be indicative of S. aureus bacteremia seeding the urinary tract. Some experts recommend obtaining blood cultures even if asymptomatic to exclude an occult bacteremia in this setting.[15] Contrary to popular belief, altered mental status and foul-smelling or cloudy urine are not true UTI symptoms.[16]
Interpreting urine tests can also be tricky. The absence of pyuria strongly argues against a UTI, while the presence of nitrite and/or leukocyte esterase is not specific for infection.[17] Misdiagnosis of UTIs is high. In one study (N = 264), 66% of females presenting to the emergency department with genitourinary symptoms were diagnosed with a UTI, but only half were correctly diagnosed.[18] Some patients tested positive for a sexually transmitted infection instead. Diagnosing UTIs in elderly patients is even more challenging, because up to 50% of females and 40% of males in long-term care have asymptomatic bacteriuria, and often pyuria as well.[17] Urine testing in this population is less helpful in diagnosing UTIs, as abnormal urine results can be expected. Thus, a urine sample should be collected only when true UTI symptoms are present, as recommended by recent guidelines.[16,19] A common indication to collect a urine sample is delirium, but other causes, such as dehydration, drug side effects, and sleep disturbances, should be ruled out before attributing the altered mental state to a UTI.[16] Regarding UTI treatment, one study found that about half of outpatient antibiotic prescriptions were consistent with guideline-recommended first-line therapy, while over 75% of prescribed durations were too long.[20]
Skin and soft tissue infections
Cellulitis and erysipelas are common bacterial infections of the skin. There is no single piece of history, physical exam finding, or lab test that can confirm the diagnosis with a high degree of accuracy.[21] Aside from a skin biopsy, which is not routinely performed, there is no way to prove that the skin or subcutaneous tissues are infected. As a result, the clinical gold standard is history and physical exam, but can any clinician provide the gold standard in diagnosing skin infections? In a randomized controlled trial (N = 175) examining the accuracy of cellulitis diagnoses where a dermatology consult was considered the gold standard, of all patients admitted to hospital with cellulitis, 30% had an alternative dermatological diagnosis such as eczema or dermatitis.[22] Patients treated by dermatologists had more rapid clinical improvement and reduced exposure to antibiotics. Similarly, in another randomized controlled trial (N = 29) of outpatients who were labeled by their primary care provider as having cellulitis, only 5 (17%) were judged to have been correctly diagnosed according to a dermatologist.[23]
Skin infections can be challenging to diagnose because there are many mimickers, also known as pseudocellulitis conditions. These include venous stasis dermatitis, contact dermatitis, eczema, deep vein thrombosis, gout, hematoma, erythema migrans, and peripheral artery disease.[21,22] When an infection is present, there are only two major pathogens to consider: beta-hemolytic streptococcus in nonpurulent infections and Staphylococcus aureus in purulent disease.[21,24] Coverage for Gram-negative organisms is indicated only in select circumstances, including moderate to severe diabetic foot infections, perineal infections, animal bite infections, aquatic-related injury, surgical site infections of the groin or axilla, severely immunocompromised patients, and necrotizing infections.[21,25]
It is expected that the choice of antibiotic should reflect the microbiological understanding of this disease. However, studies have shown that almost half of hospitalized patients receive unnecessary broad-spectrum antibiotic (most commonly piperacillin-tazobactam), vancomycin use is inappropriate in 75% of cases, up to half of patients who are eligible for oral antibiotic receive IV treatment longer than required, and over half of patients are treated longer than the guideline recommendation of 5 to 7 days.[22,26-28] When treating cellulitis, it is paramount to understand its natural progression to avoid treatment mishaps. Some clinicians might broaden the antibiotic or switch to IV therapy unnecessarily when they judge that the initial oral treatment is failing. The symptoms and signs of inflammation can worsen in the first 1 to 2 days while on effective therapy, and it can take up to 3 days before a clinical response is observed.[29] Misdiagnosing a treatment failure too early in the course of illness results in inappropriate modifications to treatment and might lead clinicians to falsely attribute the improvement to the new therapy.[30] Furthermore, clinicians often misinterpret persistent symptoms and signs of inflammation of the affected limb as evidence for ongoing infection and, as a result, extend the antibiotic treatment beyond the guideline-recommended duration. Residual inflammation at the end of treatment is normal in up to 60% of patients, and prolonging the course of antibiotic is not useful, as the bacteria are likely dead by that point.[29] Tissue repair and immune activation can persist well after the bacteria have been killed.
Conclusions
Pneumonia, urinary tract infections, and cellulitis in adult patients tend to be overdiagnosed and overtreated. Potential reasons include inadequate training in and modeling of diagnostic reasoning, the presence of cognitive biases, cultural normalization of antibiotic overuse, lack of awareness of recent evidence and guidelines, and clinical inertia. A positive test does not diagnose a disease; it is the clinician interpreting the relevant data in the right clinical context who does. Answers in medicine are rarely black and white, and operating in the grey zone demands a reasonable understanding of probabilistic rationalization and an ability to incorporate best evidence into clinical decision making. A rational approach is for clinicians to critically appraise their knowledge and skills to continually refine their competency, and thereby deliver the right amount and type of care for each patient.
hidden
This article has been peer reviewed.
 |
| This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License. |
References
1. Hastings SN, Whitson HE, Purser JL, et al. Emergency department discharge diagnosis and adverse health outcomes in older adults. J Am Geriatr Soc 2009;57:1856-1861. https://doi.org/10.1111/j.1532-5415.2009.02434.x.
2. Finley CR, Chan DS, Garrison S, et al. What are the most common conditions in primary care? Systematic review. Can Fam Physician 2018;64:832-840.
3. Ellis Simonsen SM, van Orman ER, Hatch BE, et al. Cellulitis incidence in a defined population. Epidemiol Infect 2006;134:293-299. https://doi.org/10.1017/s095026880500484X.
4. Morgan DJ, Pineles L, Owczarzak J, et al. Accuracy of practitioner estimates of probability of diagnosis before and after testing. JAMA Intern Med 2021;181:747-755. https://doi.org/10.1001/jamainternmed.2021.0269.
5. Marchello CS, Ebell MH, Dale AP, et al. Signs and symptoms that rule out community-acquired pneumonia in outpatient adults: A systematic review and meta-analysis. J Am Board Fam Med 2019;32:234-247. https://doi.org/10.3122/jabfm.2019.02.180219.
6. Simel DL, Rennie D, editors. Pneumonia, adult, community-acquired. In: The rational clinical examination: Evidence-based clinical diagnosis. Columbus, OH: McGraw-Hill Education; 2009. Accessed 4 July 2025. https://jamaevidence.mhmedical.com/content.aspx?bookid=845§ionid=61357585.
7. Laursen CB, Sloth E, Lambrechtsen J, et al. Diagnostic performance of chest X-ray for the diagnosis of community acquired pneumonia in acute admitted patients with respiratory symptoms. Scand J Trauma Resusc Emerg Med 2013;21(Suppl 2):A21. https://doi.org/10.1186/1757-7241-21-S2-A21.
8. Wattengel BA, Sellick JA, Skelly MK, et al. Outpatient antimicrobial stewardship: Targets for community-acquired pneumonia. Clin Ther 2019;41:466-476. https://doi.org/10.1016/j.clinthera.2019.01.007.
9. Torres A, Cilloniz C, Niederman MS, et al. Pneumonia. Nat Rev Dis Primers 2021;7:25. https://doi.org/10.1038/s41572-021-00259-0.
10. Jain S, Self WH, Wunderink RG, et al. Community-acquired pneumonia requiring hospitalization among US adults. N Engl J Med 2015;373:415-27. https://doi.org/10.1056/NEJMoa1500245.
11. Metlay JP, Waterer GW, Long AC, et al. Diagnosis and treatment of adults with community-acquired pneumonia. An official clinical practice guideline of the American Thoracic Society and Infectious Diseases Society of America. Am J Respir Crit Care Med 2019;200:e45-e67. https://doi.org/10.1164/rccm.201908-1581ST.
12. Baghdadi JD, Korenstein D, Pineles L, et al. Exploration of primary care clinician attitudes and cognitive characteristics associated with prescribing antibiotics for asymptomatic bacteriuria. JAMA Netw Open 2022;5:e2214268. https://doi.org/10.1001/jamanetworkopen.2022.14268.
13. Petty LA, Vaughn VM, Flanders SA, et al. Risk factors and outcomes associated with treatment of asymptomatic bacteriuria in hospitalized patients. JAMA Intern Med 2019;179:1519-1527. https://doi.org/10.1001/jamainternmed.2019.2871.
14. Aubin C. Evidence-based emergency medicine/rational clinical examination abstract. Does this woman have an acute uncomplicated urinary tract infection? Ann Emerg Med 2007;49:106-108. https://doi.org/10.1016/j.annemergmed.2006.09.022.
15. Mason CY, Sobti A, Goodman AL. Staphylococcus aureus bacteriuria: Implications and management. JAC Antimicrob Resist 2023;5:dlac123. https://doi.org/10.1093/jacamr/dlac123.
16. Association of Medical Microbiology and Infectious Disease Canada. Diagnosis of suspected urinary tract infection (UTI) in non-catheterized elderly patients in acute care setting. September 2021. Accessed 4 July 2025. https://ammi.ca/wp-content/uploads/2021/09/AC-Algorithm_Eng_8.5x11_colour.pdf.
17. Schulz L, Hoffman RJ, Pothof J, Fox B. Top ten myths regarding the diagnosis and treatment of urinary tract infections. J Emerg Med 2016;51:25-30. https://doi.org/10.1016/j.jemermed.2016.02.009.
18. Tomas ME, Getman D, Donskey CJ, Hecker MT. Overdiagnosis of urinary tract infection and underdiagnosis of sexually transmitted infection in adult women presenting to an emergency department. J Clin Microbiol 2015;53:2686-2692. https://doi.org/10.1128/JCM.00670-15.
19. Association of Medical Microbiology and Infectious Disease Canada. Five tests and treatments to question in medical microbiology and infectious diseases. Choosing Wisely Canada. Last updated June 2021. Accessed 10 September 2025. https://choosingwiselycanada.org/recommendation/medical-microbiology/.
20. Durkin MJ, Keller M, Butler AM, et al. An assessment of inappropriate antibiotic use and guideline adherence for uncomplicated urinary tract infections. Open Forum Infect Dis 2018;5:ofy198. https://doi.org/10.1093/ofid/ofy198.
21. Raff AB, Kroshinsky D. Cellulitis: A review. JAMA 2016;316:325-337. https://doi.org/10.1001/jama.2016.8825.
22. Ko LN, Garza-Mayers AC, St. John J, et al. Effect of dermatology consultation on outcomes for patients with presumed cellulitis: A randomized clinical trial. JAMA Dermatol 2018;154:529-536. https://doi.org/10.1001/jamadermatol.2017.6196.
23. Arakaki RY, Strazzula L, Woo E, Kroshinsky D. The impact of dermatology consultation on diagnostic accuracy and antibiotic use among patients with suspected cellulitis seen at outpatient internal medicine offices: A randomized clinical trial. JAMA Dermatol 2014;150:1056-1061. https://doi.org/10.1001/jamadermatol.2014.1085.
24. Lee CY, Tsai HC, Kunin CM, et al. Clinical and microbiological characteristics of purulent and non-purulent cellulitis in hospitalized Taiwanese adults in the era of community-associated methicillin-resistant Staphylococcus aureus. BMC Infect Dis 2015;15:311. https://doi.org/10.1186/s12879-015-1064-z.
25. Stevens DL, Bisno AL, Chambers HF, et al. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the Infectious Diseases Society of America. Clin Infect Dis 2014;59:e10-52. https://doi.org/10.1093/cid/ciu296.
26. Brindle R, Williams OM, Barton E, Featherstone P. Assessment of antibiotic treatment of cellulitis and erysipelas: A systematic review and meta-analysis. JAMA Dermatol 2019;155:1033-1040. https://doi.org/10.1001/jamadermatol.2019.0884.
27. Cross ELA, Jordan H, Godfrey R, et al. Route and duration of antibiotic therapy in acute cellulitis: A systematic review and meta-analysis of the effectiveness and harms of antibiotic treatment. J Infect 2020;81:521-531. https://doi.org/10.1016/j.jinf.2020.07.030.
28. Almulhim AS, Alotaibi FM. Comparison of broad-spectrum antibiotics and narrow-spectrum antibiotics in the treatment of lower extremity cellulitis. Int J Health Sci (Qassim) 2018;12:3-7.
29. Bruun T, Oppegaard O, Hufthammer KO, et al. Early response in cellulitis: A prospective study of dynamics and predictors. Clin Infect Dis 2016;63:1034-1041. https://doi.org/10.1093/cid/ciw463.
30. Brindle R, Williams OM, Davies P, et al. Adjunctive clindamycin for cellulitis: A clinical trial comparing flucloxacillin with or without clindamycin for the treatment of limb cellulitis. BMJ Open 2017;7:e013260. https://doi.org/10.1136/bmjopen-2016-013260.
hidden
Dr Wong is a clinical assistant professor in the Division of Infectious Diseases, Department of Medicine, University of British Columbia, and an infectious diseases consultant at Royal Columbian Hospital and Eagle Ridge Hospital.
Corresponding author: Dr Davie Wong, davie.wong@fraserhealth.ca.