Iliocaval reconstruction for the treatment of postthrombotic syndrome
ABSTRACT: Postthrombotic syndrome is the result of chronic venous obstruction causing symptoms of pain, heaviness, cramping, edema, pruritus, and venous claudication. We present the case of a patient with severe postthrombotic syndrome as a result of chronic deep vein thrombosis of the inferior vena cava, bilateral iliac veins, and infrainguinal deep venous system, secondary to compression by a large conglomerate of lymph nodes due to non-Hodgkin lymphoma. The patient was successfully treated with a complex venous reconstruction procedure involving recanalization, mechanical thrombectomy, and stenting. This case highlights evolving techniques for the management of postthrombotic syndrome.
The case of a complex venous reconstruction procedure involving recanalization, mechanical thrombectomy, and stenting highlights evolving techniques for the management of postthrombotic syndrome.
Background
Deep vein thrombosis is the development of thrombus in the deep venous system, typically in the legs. Conventional first-line therapy is management with oral anticoagulants or occasionally parenteral anticoagulants in specific populations. Up to 50% of patients with deep vein thrombosis have residual vascular obstruction despite adequate anticoagulation.[1-4] Residual vascular obstruction is associated with adverse events, which include a 3-times-higher risk of death, a 3-times-higher risk of recurrent deep vein thrombosis, and a greater risk of developing postthrombotic syndrome.[1-3,5] Up to 50% of patients with deep vein thrombosis develop postthrombotic syndrome;[6] symptoms include lower extremity pain, heaviness, cramping, edema, pruritus, and venous claudication (including venous leg ulcers).[7] These symptoms can be severely debilitating: patients with severe postthrombotic syndrome have a quality of life that is comparable to that of patients with congestive heart failure or cancer, and 90% of patients are unable to work 10 years after their diagnosis.[8,9] Thus, it is imperative to recognize and manage the disease early. As endovascular equipment and techniques have evolved, more procedural options have become available as adjuncts to standard treatment.
We present a case report of endovascular iliocaval reconstruction for the management of a patient who presented with severe postthrombotic syndrome secondary to a deep vein thrombosis due to a large conglomerate mass in the context of stage 4 non-Hodgkin lymphoma. This case report presents an emerging technique in the management of an underrecognized pathology and underscores the importance of multidisciplinary collaboration in the management of complex pathology.
Case description
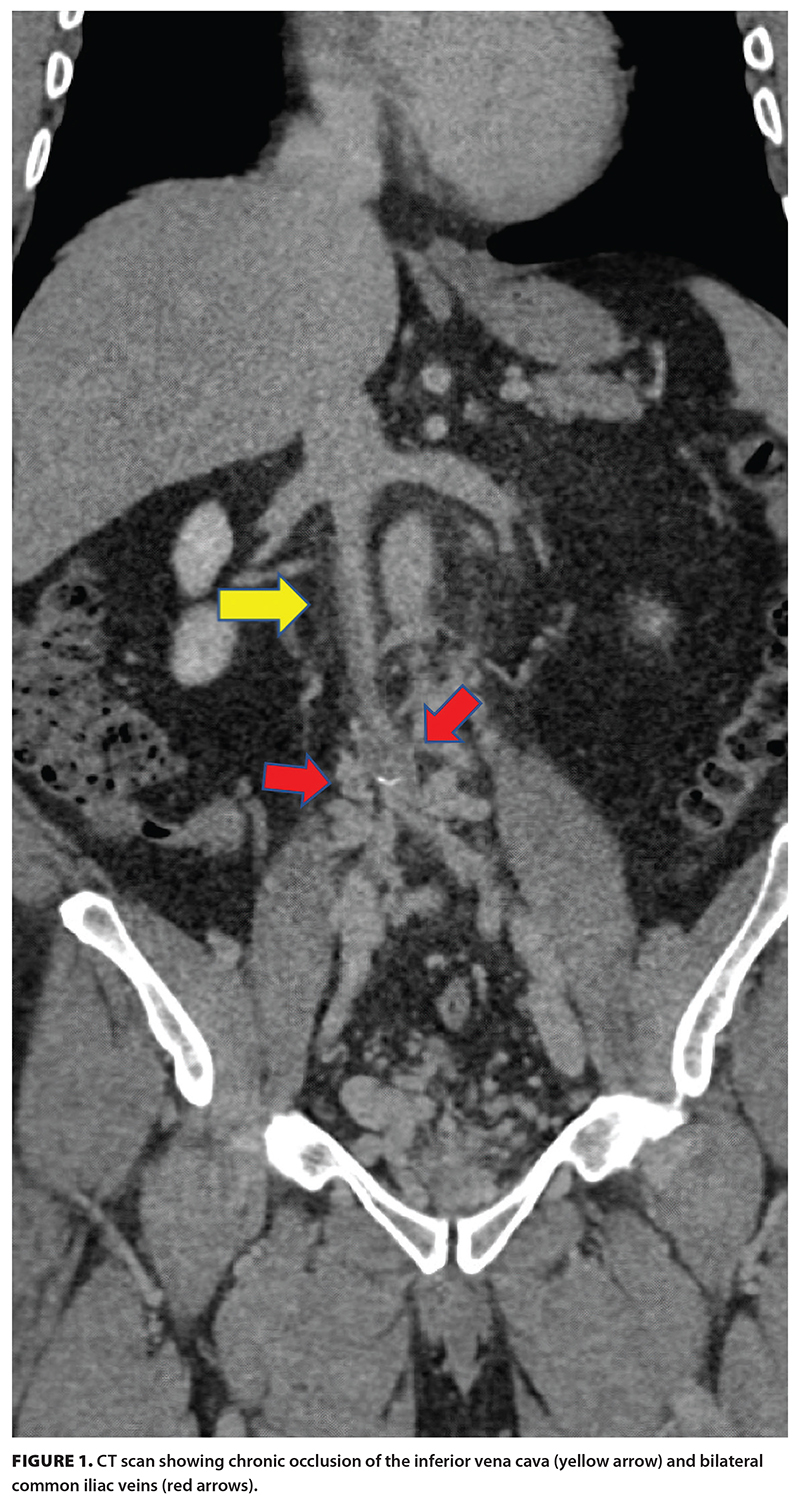 A 46-year-old patient was diagnosed with stage 4 non-Hodgkin lymphoma after presenting with a large nodal conglomerate retroperitoneal mass. They subsequently underwent an extended course of chemoimmunotherapy for the treatment of their lymphoma. However, the retroperitoneal mass caused compression on adjacent vasculature and resulted in occlusive thrombus formation within the deep venous system, extending from the inferior vena cava to the ankles [Figure 1]. This required therapeutically dosed anticoagulation, and although the lymphoma went into remission, the thrombus remained. The patient developed significant postthrombotic syndrome as a result of this residual thrombus and experienced symptoms of chronic swelling, pain, heaviness, and, most importantly, venous claudication. These symptoms significantly impacted their quality of life, because daily activities such as walking short distances and exercising were intolerable. Their symptom severity correlated with a severe Villalta score of 22.
A 46-year-old patient was diagnosed with stage 4 non-Hodgkin lymphoma after presenting with a large nodal conglomerate retroperitoneal mass. They subsequently underwent an extended course of chemoimmunotherapy for the treatment of their lymphoma. However, the retroperitoneal mass caused compression on adjacent vasculature and resulted in occlusive thrombus formation within the deep venous system, extending from the inferior vena cava to the ankles [Figure 1]. This required therapeutically dosed anticoagulation, and although the lymphoma went into remission, the thrombus remained. The patient developed significant postthrombotic syndrome as a result of this residual thrombus and experienced symptoms of chronic swelling, pain, heaviness, and, most importantly, venous claudication. These symptoms significantly impacted their quality of life, because daily activities such as walking short distances and exercising were intolerable. Their symptom severity correlated with a severe Villalta score of 22.
The patient’s CT scan showed chronic occlusion of the infrarenal inferior vena cava and bilateral iliac system, with extensive pelvic, paralumbar, retroperitoneal, and abdominal wall venous collaterals. The patient’s hematologist consulted interventional radiology for endovascular management options. After a detailed discussion of the proposed procedural intervention, the importance of postprocedural antithrombotics, and long-term follow-up, the patient was keen to proceed.
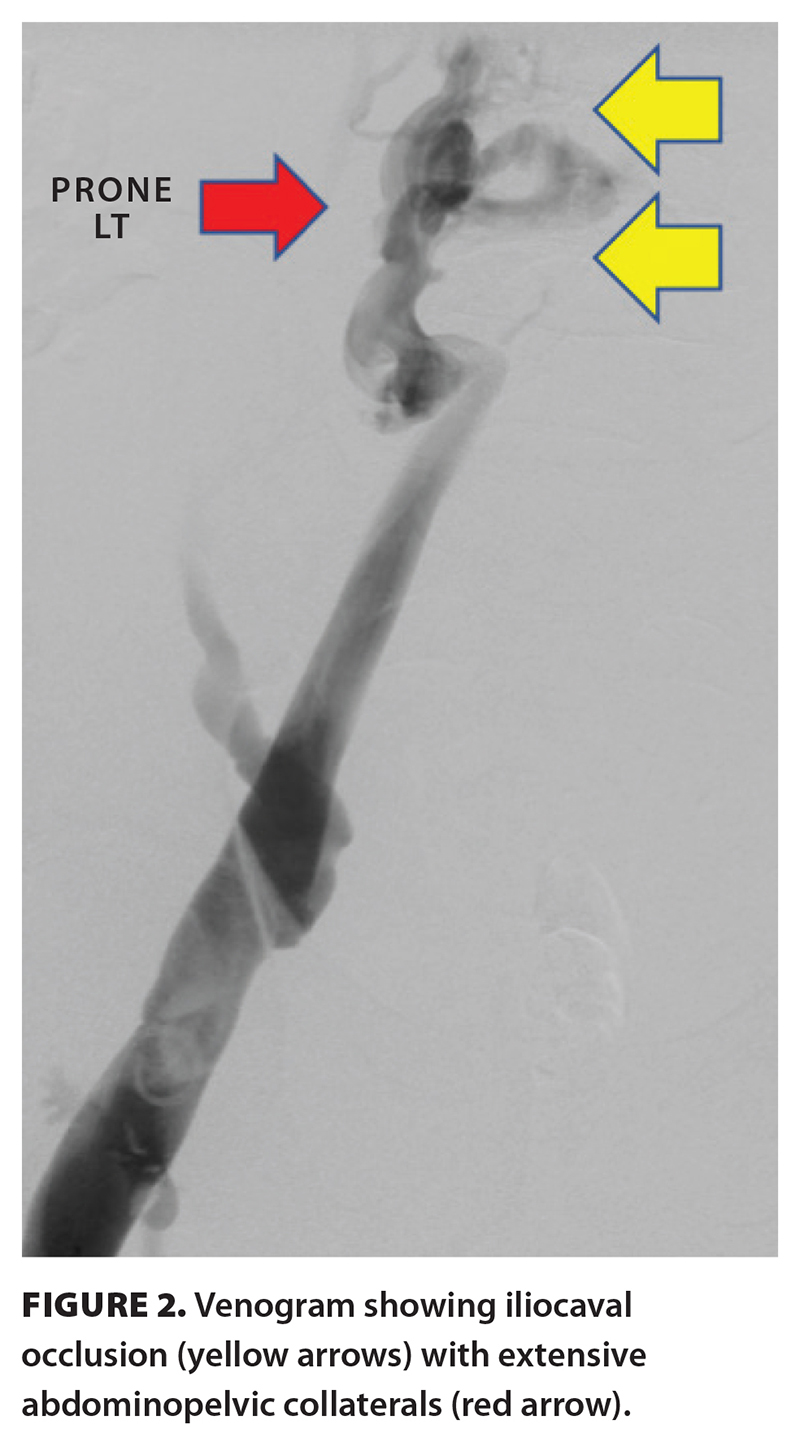 In December 2024, the patient was brought to the angiography suite at Vancouver General Hospital and placed under general anesthesia. Under fluoroscopy, catheter-directed venography showed chronic right lower extremity deep vein thrombosis and iliocaval occlusion with extensive venous collaterals [Figure 2]. Mechanical thrombectomy of the right iliofemoral deep vein thrombosis was performed with subsequent recanalization of the occluded iliocaval veins and venous reconstruction with stents extending from the infrarenal inferior vena cava into the bilateral external iliac veins. On day 1 postprocedure, check venography was performed with augmentation of the stent constructs through balloon dilation, which resulted in widely patent stents and antegrade flow with no residual filling of the collaterals. The patient was discharged 1 day later with daily low-dose aspirin and therapeutically dosed low-molecular-weight heparin.
In December 2024, the patient was brought to the angiography suite at Vancouver General Hospital and placed under general anesthesia. Under fluoroscopy, catheter-directed venography showed chronic right lower extremity deep vein thrombosis and iliocaval occlusion with extensive venous collaterals [Figure 2]. Mechanical thrombectomy of the right iliofemoral deep vein thrombosis was performed with subsequent recanalization of the occluded iliocaval veins and venous reconstruction with stents extending from the infrarenal inferior vena cava into the bilateral external iliac veins. On day 1 postprocedure, check venography was performed with augmentation of the stent constructs through balloon dilation, which resulted in widely patent stents and antegrade flow with no residual filling of the collaterals. The patient was discharged 1 day later with daily low-dose aspirin and therapeutically dosed low-molecular-weight heparin.
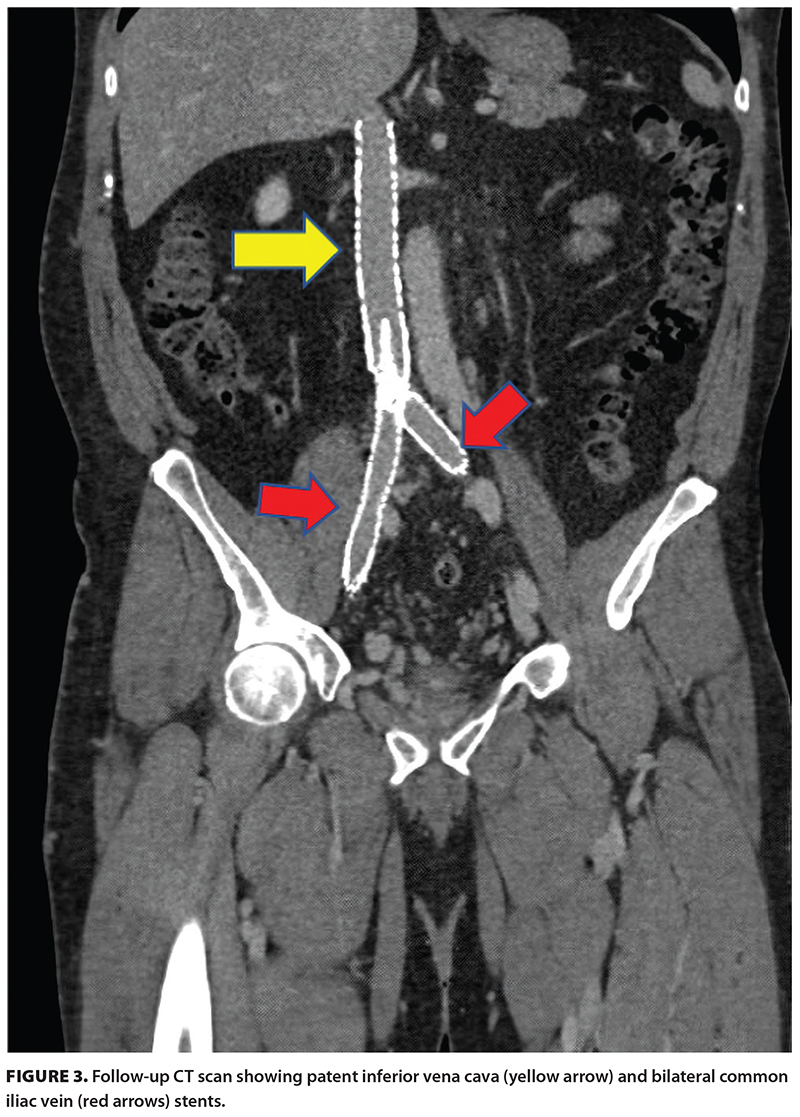 The patient lost 13 kg in 3 weeks due to improvement of their venous hypertension and resultant reduction in edema. They were able to advance from walking with a cane to hiking and biking without assistance. They returned to a fit and active lifestyle, nearing their prelymphoma diagnosis status, with a Villalta score of 1. Follow-up assessment 1 year postintervention demonstrated ongoing patency of the stents [Figure 3]. The patient is managed by our multidisciplinary hematology and interventional radiology team and continues to do very well, with no lifestyle limitations.
The patient lost 13 kg in 3 weeks due to improvement of their venous hypertension and resultant reduction in edema. They were able to advance from walking with a cane to hiking and biking without assistance. They returned to a fit and active lifestyle, nearing their prelymphoma diagnosis status, with a Villalta score of 1. Follow-up assessment 1 year postintervention demonstrated ongoing patency of the stents [Figure 3]. The patient is managed by our multidisciplinary hematology and interventional radiology team and continues to do very well, with no lifestyle limitations.
Discussion
With an estimated annual incidence of deep vein thrombosis being approximately 100 cases per 100 000 person years and postthrombotic syndrome developing in up to 50% of those patients, postthrombotic syndrome is an underdiagnosed condition.[10-12] Although there are a few objective measurement tools for grading the severity of postthrombotic syndrome, including the Villalta score, they all have a glaring omission in that they fail to measure venous claudication, which can cause significant morbidity. Currently, the mainstays of postthrombotic syndrome management are lifestyle modifications (e.g., leg elevation, weight loss, exercise) and compression therapy.[13,14] However, there is growing evidence for adjunctive endovascular management of postthrombotic syndrome in patients with lifestyle-limiting disease.[15]
Endovascular reconstruction using recanalization techniques, balloon angioplasty, thrombectomy, and stenting are viable management options for patients with postthrombotic syndrome secondary to iliocaval obstruction; studies have demonstrated low complication rates, high midterm patency rates, and significant clinical improvement.[16,17] For advanced disease, venoplasty and stenting in patients with iliofemoral obstruction resulted in healing of venous stasis-related ulcers in 55% of cases.[18] Similarly, the ACCESS PTS study demonstrated high success rates with the use of ultrasound-accelerated thrombolysis and venoplasty for recanalization of chronic venous obstructions and improved quality of life in patients during 1 year of follow-up.[19] Despite the evidence supporting the additive value of endovascular management of postthrombotic syndrome, long-term management of the condition still hinges on a multidisciplinary approach that ensures appropriate patient selection, management of comorbidities, wound care, adherence to anticoagulation/antiplatelet therapy, and comprehensive collaborative follow-up.[20]
Our patient had developed severe postthrombotic syndrome secondary to a central venous outflow obstruction due to compression from their large conglomerate retroperitoneal nodal disease. This led to extensive thrombosis, atresia of native vasculature, and extensive collateral vessel formation. By emphasizing a patient-centred and multidisciplinary approach and having discussions with the patient and their family, the procedure was a resounding radiographic and clinical success, with profound symptom improvement. Our patient remains on aspirin and rivaroxaban. Through our established venous thromboembolism pathway, we follow patients with this pathology longitudinally. Our patient’s care highlights the importance of appropriate patient selection and the use of a multidisciplinary approach for the management of complex venous pathology.
Conclusions
Endovascular techniques are a promising and evolving management option for postthrombotic syndrome.
Funding
None declared.
Competing interests
None declared.
Consent statement
Written informed consent for publication of this case report was obtained from the patient.
This article has been peer reviewed.

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
References
1. Young L, Ockelford P, Milne D, et al. Post-treatment residual thrombus increases the risk of recurrent deep vein thrombosis and mortality. J Thromb Haemost 2006;4:1919-1924. https://doi.org/10.1111/j.1538-7836.2006.02120.x.
2. Yoo T, Aggarwal R, Wang T-F, et al. Presence and degree of residual venous obstruction on serial duplex imaging is associated with increased risk of recurrence and progression of infrainguinal lower extremity deep venous thrombosis. J Vasc Surg Venous Lymphat Disord 2018;6:575-583.e1. https://doi.org/10.1016/j.jvsv.2017.12.059.
3. Dronkers CEA, Mol GC, Maraziti G, et al. Predicting post-thrombotic syndrome with ultrasonographic follow-up after deep vein thrombosis: A systematic review and meta-analysis. Thromb Haemost 2018;118:1428-1438. https://doi.org/10.1055/s-0038-1666859.
4. Prandoni P, Lensing AWA, Prins MH, et al. Residual venous thrombosis as a predictive factor of re-current venous thromboembolism. Ann Intern Med 2002;137:955-960. https://doi.org/10.7326/0003-4819-137-12-200212170-00008.
5. Avgerinos ED, Saadeddin Z, Abou Ali AN, et al. Outcomes and predictors of failure of iliac vein stenting after catheter-directed thrombolysis for acute iliofemoral thrombosis. J Vasc Surg Venous Lymphat Disord 2019;7:153-161. https://doi.org/10.1016/j.jvsv.2018.08.014.
6. Centers for Disease Control and Prevention. Impact of blood clots on the United States infographic. 15 May 2024. Accessed 17 October 2024. www.cdc.gov/blood-clots/toolkit/impact-of-blood-clots.html.
7. Galanaud J-P, Bertoletti L, Amitrano M, et al. Predictors of post-thrombotic ulcer after acute DVT: The RIETE registry. Thromb Haemost 2018;118:320-328. https://doi.org/10.1160/TH17-08-0598.
8. Kahn SR, Ginsberg JS. Relationship between deep venous thrombosis and the postthrombotic syndrome. Arch Intern Med 2004;164:17-26. https://doi.org/10.1001/archinte.164.1.17.
9. Office of the Surgeon General (US); National Heart, Lung, and Blood Institute (US). The Surgeon General’s call to action to prevent deep vein thrombosis and pulmonary embolism. Rockville, MD: Office of the Surgeon General (US); 2008. Accessed 17 October 2024. www.ncbi.nlm.nih.gov/books/NBK44178/.
10. White RH. The epidemiology of venous thromboembolism. Circulation 2003;107(23 Suppl 1):I4-I8. https://doi.org/10.1161/01.CIR.0000078468.11849.66.
11. Chopard R, Albertsen IE, Piazza G. Diagnosis and treatment of lower extremity venous thromboembolism: A review. JAMA 2020;324:1765-1776. https://doi.org/10.1001/jama.2020.17272.
12. Kahn SR, Galanaud J-P, Vedantham S, Ginsberg JS. Guidance for the prevention and treatment of the post-thrombotic syndrome. J Thromb Thrombolysis 2016;41:144-153. https://doi.org/10.1007/s11239-015-1312-5.
13. Azirar S, Appelen D, Prins MH, et al. Compression therapy for treating post-thrombotic syndrome. Cochrane Database Syst Rev 2019;(9):CD004177. https://doi.org/10.1002/14651858.CD004177.pub2.
14. Kahn SR. The post-thrombotic syndrome. Hemat-ology Am Soc Hematol Educ Program 2016;2016:413-418. https://doi.org/10.1182/asheducation-2016.1.413.
15. Chaitidis N, Kokkinidis DG, Papadopoulou Z, et al. Management of post-thrombotic syndrome: A comprehensive review. Curr Pharm Des 2022;28:550-559. https://doi.org/10.2174/1381612828666220131094655.
16. David A, Thony F, Del Giudice C, et al. Short- and mid-term outcomes of endovascular stenting for the treatment of post-thrombotic syndrome due to iliofemoral and caval occlusive disease: A multi-centric study from the French Society of Diagnostic and Interventional Cardiovascular Imaging (SFICV). Cardiovasc Intervent Radiol 2022;45:162-171. https://doi.org/10.1007/s00270-021-03038-2.
17. Rossi FH, Kambara AM, Izukawa NM, et al. Randomized double-blinded study comparing medical treatment versus iliac vein stenting in chronic venous disease. J Vasc Surg Venous Lymphat Disord 2018;6:183-191. https://doi.org/10.1016/j.jvsv.2017.11.003.
18. Neglén P, Hollis KC, Olivier J, Raju S. Stenting of the venous outflow in chronic venous disease: Long-term stent-related outcome, clinical, and hemodynamic result. J Vasc Surg 2007;46:979-990. https://doi.org/10.1016/j.jvs.2007.06.046.
19. Garcia MJ, Sterling KM, Kahn SR, et al. Ultrasound-accelerated thrombolysis and venoplasty for the treatment of the postthrombotic syndrome: Results of the ACCESS PTS study. J Am Heart Assoc 2020;9:e013398. https://doi.org/10.1161/JAHA.119.013398.
20. Li W, Vedantham S, Jaffer FA, et al. Revisiting the open vein hypothesis to reduce the postthrombotic syndrome: Implications for multidisciplinary care and research: A scientific statement from the American Heart Association. Circulation 2025;151. https://doi.org/10.1161/CIR.0000000000001330.
Dr Anand Dhatt is a resident physician in the Department of Radiology, Faculty of Medicine, University of British Columbia. Dr Heran is a staff interventional neuroradiologist in the Department of Radiology at Vancouver General Hospital. Dr Ho is a staff interventional radiologist in the Department of Radiology at VGH. Dr de Korompay is a staff interventional radiologist in the Department of Radiology at Kelowna General Hospital. Dr Min is a staff interventional radiologist in the Department of Radiology at St. Paul’s Hospital. Dr Zypchen is a staff hematologist in the Department of Hematology at VGH. Dr Peterson is a staff hematologist in the Department of Hematology at VGH. Dr Lai is a staff hematologist in the Department of Hematology at VGH and a clinical associate professor at the Centre for Blood Research, UBC. Dr Ravjot Dhatt is a staff interventional radiologist in the Department of Radiology at VGH.
Corresponding author: Dr Ravjot Dhatt, Ravjot.Dhatt@vch.ca.
